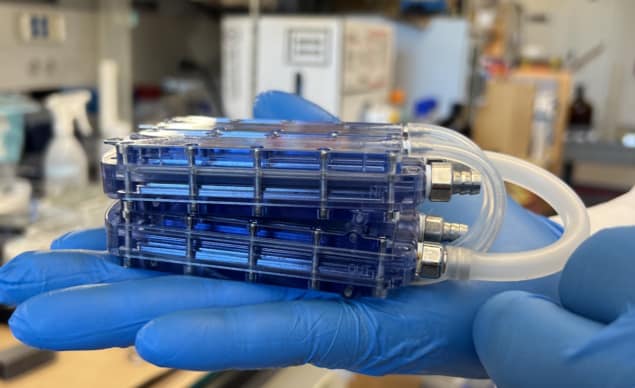
End-stage renal disease, when the kidneys can no longer support the body’s needs, is best treated via kidney transplantation. The supply of donor organs is limited, however, and many patients must rely on dialysis instead. And while a kidney transplant provides excellent outcomes, it requires the patient to take lifelong immunosuppression drugs, which cause their own health complications. To address these issues, researchers are hoping to develop an implantable bioartificial kidney to treat kidney failure.
Researchers at UC San Francisco (UCSF) are working to create such a device. In their latest study, reported in Nature Communications, they demonstrate that kidney cells housed in an implantable bioreactor could survive inside a pig and mimic several important kidney functions. Crucially, the implanted device did not provoke an immune reaction. The research is part of The Kidney Project, jointly headed by UCSF’s Shuvo Roy and William Fissell of Vanderbilt University Medical Center.
As an initial step towards an implantable bioartificial kidney, Roy, Fissel and colleagues created a cell-containing bioreactor that replicates key functions of the renal tubule, which includes delivering oxygen and nutrients to kidney cells while protecting them from recipient immune cells that could cause rejection. To achieve this immunoprotection, the team engineered a thin (less than 1 μm) silicon nanopore membrane (SNM) from silicon wafers containing nanoscale slit pores.
The researchers built a prototype bioreactor housing an SNM with 10-nm-wide pores, with human renal epithelial cells (HRECs) cultured on acrylic inserts either side of the SNM. To test whether the SNM provides an immunoprotective barrier in vitro, they exposed one side of the SNM to the proinflammatory cytokine TNF-α.
Six hours after exposure, levels of TNF-α in the compartment protected by the SNM were negligible, demonstrating that the 10-nm-wide pores prevent the passage of large cytokines (and thus also larger immune system components such as T cells and antibodies). Cells on the side directly exposed to TNF-α had less than 50% viability, while those sealed by the SNM maintained a high viability of nearly 90%.
It’s also important that an implant does not cause blood clots, which can form on surfaces in contact with blood and lead to device failure and serious complications for the patient. To avoid this, the researchers employed computational fluid dynamics to optimize the geometry of the U-shaped blood flow path though the bioreactor.
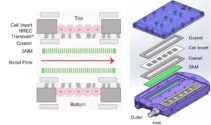
They used this design to create an implantable bioreactor containing four SNMs in a polycarbonate housing, aligned with the top and bottom surfaces of the blood channels. They placed acrylic inserts seeded with confluent HRECs on the opposite sides of each SNM, and used stainless steel connectors to transition the blood between the SNMs and PTFE vascular grafts.
To test the biocompatibility of these bioreactor prototypes, the team assembled and surgically implanted devices into five healthy pigs on dual antiplatelet therapy (without immunosuppression). The bioreactors connected to the animals’ vasculature via the PTFE grafts. No complications related to the devices or the surgery occurred and the animals remained healthy during the throughout the three- or seven-day experiments (the time in which hyperacute rejection would typically occur), with no sign of rejection.
The researchers evaluated whether implanting bioreactors containing xenogeneic human cells provoked an immune reaction in the pigs. Examining 13 common inflammatory biomarkers revealed an increase in some cytokines two days after implant, as expected due to postoperative inflammation. But seven days after implant, all cytokine levels had decreased to a state of minimal inflammation. This finding is in contrast to the vigorous response expected in a typical xenograft model with an immunocompetent recipient.
OCT could help expand the kidney donor pool
After three or seven days, the researchers removed the bioreactors from the animals for evaluation. They found that the explanted SNM was intact, with minimal cell and protein attachment and no blood clots formed in the device – an important step in demonstrating its safety. In addition, the encapsulated HREC cells remained more than 90% viable, with no evidence of cell detachment and maintenance of tight intercellular junctions.
“We needed to prove that a functional bioreactor will not require immunosuppressant drugs, and we did,” says Roy in a press statement. “We had no complications and can now iterate up, reaching for the whole panel of kidney functions at the human scale.”
- SEO Powered Content & PR Distribution. Get Amplified Today.
- PlatoData.Network Vertical Generative Ai. Empower Yourself. Access Here.
- PlatoAiStream. Web3 Intelligence. Knowledge Amplified. Access Here.
- PlatoESG. Automotive / EVs, Carbon, CleanTech, Energy, Environment, Solar, Waste Management. Access Here.
- PlatoHealth. Biotech and Clinical Trials Intelligence. Access Here.
- ChartPrime. Elevate your Trading Game with ChartPrime. Access Here.
- BlockOffsets. Modernizing Environmental Offset Ownership. Access Here.
- Source: https://physicsworld.com/a/implantable-bioartificial-kidney-aims-to-free-patients-from-dialysis/